CMV Infection After Liver Transplant is strongly associated with morbidity, graft dysfunction, rejection, and mortality if not recognised and managed promptly.
While universal prophylaxis or preemptive monitoring has reduced incidence, late-onset CMV disease remains an important post-liver transplant concern.
Third week after a successful liver transplant, Mr Charan—an individual transplanted for alcohol-related liver disease—was recovering steadily in the intensive care unit. His initial postoperative course was complicated by an intra-abdominal infection, requiring antibiotics and a temporary reduction in immunosuppression.
As the infection settled, immunosuppression was carefully re-escalated. Within days, however, a new concern emerged. Liver enzymes, which had been improving, began to rise again. There was no obvious surgical cause. Vascular flow was intact. Biliary imaging was unremarkable.
The question arose—was this acute rejection, drug-induced injury, or something less obvious?
Given the timing and clinical context, cytomegalovirus (CMV) hepatitis was suspected. Blood was sent for CMV DNA quantification, and antiviral therapy was initiated while awaiting results. Over the following days, the clinical picture became clearer.
This scenario is not uncommon in liver transplant practice. CMV infection can present subtly, mimic rejection, or complicate an already complex postoperative course. Understanding when to suspect CMV, how to interpret viral load results, and when to initiate treatment is critical to protecting both graft and patient.
This blog explores CMV infection after liver transplantation—its timing, risk factors, diagnostic thresholds, and evidence-based management—based on the most recent international guidelines and transplant literature.
1. Understanding CMV After Liver Transplant
What is CMV Infection vs CMV Disease?
- CMV infection: Detection of CMV replication (usually by quantitative PCR) with or without symptoms.
- CMV disease: CMV infection with clinical symptoms — fever, malaise, leukopenia, organ involvement (GI, liver, lungs, retina).
Timing of CMV Events
CMV typically occurs:
- Early post-transplant (0–3 months)
— Often related to immunosuppression and lack of prior immunity. - Late onset (>3 months)
— May be triggered after prophylaxis cessation or increased immunosuppression (e.g., rejection therapy).
2. CMV After Liver Transplant: Clinical Presentations: What to Watch For
Presentation can be variable depending on the timing and severity.
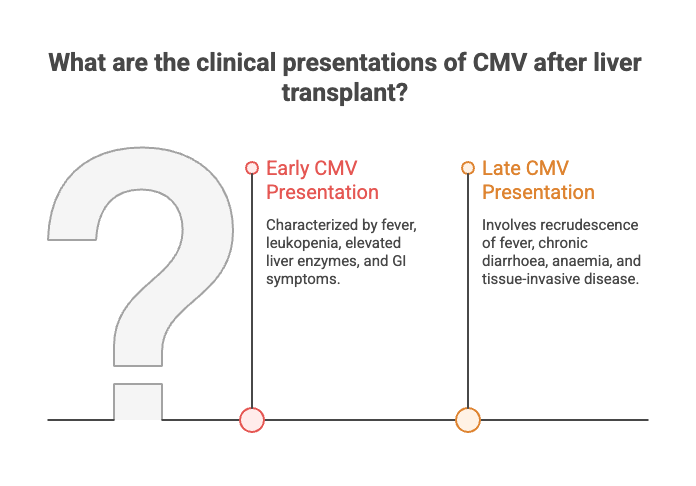
Early CMV Presentation
- Fever and constitutional symptoms
- Leukopenia and cytopenias
- Elevated liver enzymes
- GI symptoms: colitis, nausea, abdominal pain
- Possible pneumonitis or hepatitis
Late CMV Presentation
- Recrudescence of fever after stopping prophylaxis
- Chronic diarrhoea/weight loss
- Anaemia or bone marrow suppression
- Tissue-invasive disease (diagnosed on biopsy)
Importantly, tissue-invasive disease may occur even without high blood viral load — histology may be needed for definitive diagnosis.
3. Major Risk Factors for CMV After Liver Transplant
The probability of CMV infection or disease depends on several factors:
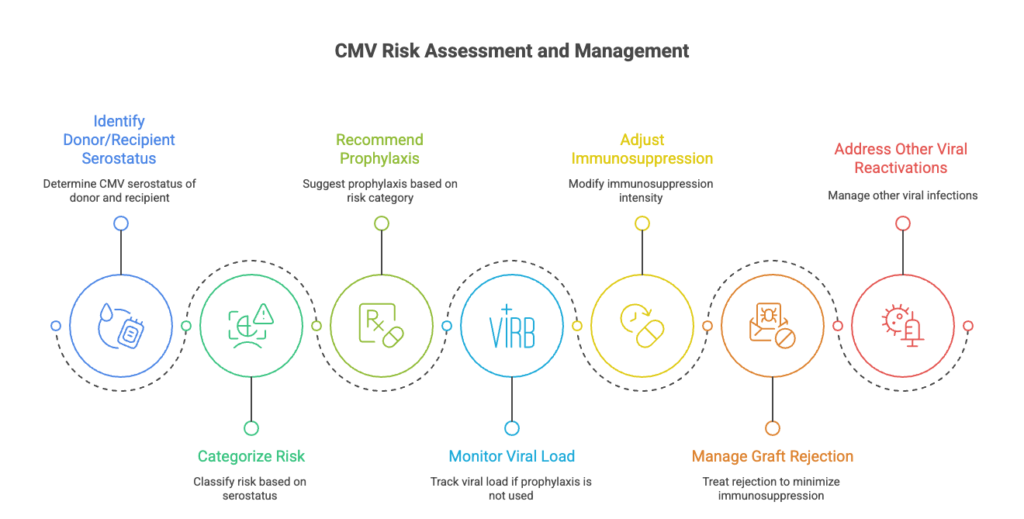
A. Donor/Recipient Serostatus
| CMV Serostatus (D/R) | Risk Category | Prophylaxis Recommendation | Comments |
|---|---|---|---|
| D+/R– | High risk | Valganciclovir prophylaxis 3–6 months | Highest risk of CMV disease; consider longer prophylaxis in high immune suppression settings. |
| D+/R+ | Intermediate risk | Valganciclovir prophylaxis ~3 months or pre-emptive monitoring | Moderate risk; prophylaxis is preferred, but a preemptive strategy is acceptable with close monitoring. |
| D–/R+ | Intermediate/low | Consider prophylaxis ~3 months or monitoring | Lower risk than D+/R+ but still may benefit from targeted prophylaxis. |
| D–/R– | Low risk | No universal prophylaxis; consider monitoring | Only monitor if clinically indicated; lowest CMV disease incidence. |
- Highest risk: Donor CMV positive / Recipient CMV negative (D+/R–)
- Moderate risk: D+/R+, D–/R+
- Lowest risk: D–/R–
B. Immunosuppression Intensity
- Anti-lymphocyte agents (e.g., ATG, alemtuzumab)
- High-dose steroids
- Mycophenolate mofetil favoured over mTOR inhibitors in some analyses
To understand the role of immunosuppressive drugs after transplant, read our detailed guide in the Know Your Medicines blog.
- Treated rejection increases risk through augmented immunosuppression.
D. Other viral reactivations can also predispose to CMV disease.
4. Viral Load Monitoring and Thresholds

📊 CMV DNAemia Thresholds (Consensus & Practical Ranges)
Important principle: There is no single universal threshold defined across all centres; thresholds are assay- and risk-dependent. Guidelines recommend weekly monitoring with locally validated viral load cut-offs, and decisions should integrate clinical context and kinetics rather than a strict absolute number alone.
| Risk Setting / Lab Measure | Practical Threshold (IU/mL) | Usual Interpretation / Action |
|---|---|---|
| Low risk / Routine monitoring | Detectable but < ~1000–3000 IU/mL | Often observe and recheck; consider kinetics before treatment. |
| Rising or intermediate load (~2000–5000 IU/mL) | ~2000–5000 IU/mL | Many centres consider this pre-emptive therapy threshold in intermediate risk. |
| Higher levels (>5000–10,000 IU/mL) | ≥ ~5000–10,000 IU/mL | Stronger indication to initiate therapy in most settings. |
| Very high or symptomatic | Varies by assay | Treat regardless of number; correlate with symptoms & risk factors. |
When CMV DNA <1,000 IU/mL SHOULD Be Treated
Even low-level DNAemia may require treatment if any of the following are present:
- High-risk serostatus (D+/R–)
- Compatible clinical features (unexplained fever, cytopenias, rising liver enzymes)
- Rapidly rising viral load on serial tests
- Recent high-dose steroids or anti-lymphocyte therapy
- Suspected tissue-invasive CMV (e.g., hepatitis, colitis)
In these situations, clinical judgment overrides the absolute viral load number.
5. Prevention: Prophylaxis vs Preemptive Monitoring
Antiviral Prophylaxis for CMV After Liver Transplant
Recommended especially in high-risk patients (D+/R–):
- Valganciclovir or intravenous ganciclovir started early post-transplant
- Duration: typically 3–6 months, sometimes longer in high-risk settings.
Pros: Lower early disease rates
Cons: Late-onset disease after stopping therapy; drug toxicity
Preemptive Therapy for CMV After Liver Transplant
- Routine monitoring (e.g., weekly PCR for 12–16 weeks)
- Start antiviral only if viral load exceeds local threshold
- Reduced drug exposure vs universal prophylaxis
Both strategies are valid but may be tailored to centre practice and patient risk.
6. Treatment for CMV After Liver Transplant: What Drugs & How Long?

First-Line Antivirals
- Valganciclovir (oral) — preferred in mild to moderate cases
- Ganciclovir (IV) — recommended for severe disease, GI involvement, poor oral absorption
Treatment Principles
- Continue antiviral therapy until:
- Symptoms resolve
- CMV DNA is undetectable or below a pre-defined lab threshold
- Minimum of ~2 weeks of therapy
- Weekly PCR monitoring is recommended during therapy.
Second-Line / Resistant CMV
- If virologic failure occurs after 2–3 weeks, consider testing for resistance.
- Alternative agents may include foscarnet or maribavir in resistant cases — guided by genotypic data.
7. Follow-Up After Treatment
- Weekly PCR monitoring until clearance
- Consider secondary prophylaxis in recurrent disease or very high-risk patients.
- Monitor for bone marrow suppression and renal toxicity from antiviral medications.
8. Practical Clinical Tips
✔ Always risk-stratify patients pre-transplant based on donor/recipient serostatus.
✔ Use prophylaxis for high-risk patients, but maintain vigilant post-prophylaxis monitoring.
✔ Low-level viremia warrants close observation and repeat testing rather than immediate full treatment — especially if asymptomatic.
✔ Adjust immunosuppression where possible to aid immune response without risking rejection.
Conclusion
CMV after liver transplantation remains a significant challenge due to its variable presentation, risk of late onset, and need for tailored treatment. Combining risk stratification, vigilant monitoring, prophylaxis/preemptive strategies, and evidence-based treatment optimises transplant outcomes in line with international practice.